Study Reveals How 98 Percent of Plastics at Sea Go Missing Each Year

Trillions of plastic fragments are afloat at sea, which cause large “garbage patches” to form in rotating ocean currents called subtropical gyres. As a result, impacts on ocean life are increasing and affecting organisms from large mammals to bacteria at the base of the ocean food web. Despite this immense accumulation of plastics at sea, it only accounts for 1 to 2 percent of plastic debris inputs to the ocean. The fate of this missing plastic and its impact on marine life remains largely unknown.
It appears that sunlight-driven photoreactions could be an important sink of buoyant plastics at sea. Sunlight also may have a role in reducing plastics to sizes below those captured by oceanic studies. This theory could partly explain how more than 98 percent of the plastics entering the oceans go missing every year. However, direct, experimental evidence for the photochemical degradation of marine plastics remains rare.
A team of scientists from ∑¨«—÷±≤•‚Äôs Harbor Branch Oceanographic Institute, and conducted a unique study to help elucidate the mystery of missing plastic fragments at sea. Their work provides novel insight regarding the removal mechanisms and potential lifetimes of a select few microplastics.¬Ý
For the study, published in the , researchers selected plastic polymers prevalently found on the ocean surface and irradiated them using a solar simulator system. The samples were irradiated under simulated sunlight for approximately two months to capture the kinetics of plastic dissolution. Twenty-four hours was the equivalent of about one solar day of photochemical exposure in the subtropical ocean gyre surface waters. To assess the physical and chemical photodegradation of these plastics, researchers used optical microscopy, electron microscopy, and Fourier transform infrared (FT-IR) spectroscopy.¬Ý
Results showed that simulated sunlight increased the amount of dissolved carbon in the water and made those tiny plastic particles tinier. It also fragmented, oxidized and altered the color of the irradiated polymers. Rates of removal depended upon polymer chemistry. Engineered polymer solutions (recycled plastics) degraded more rapidly than polypropylene (e.g. consumer packaging) and polyethylene (e.g. plastic bags, plastic films, and containers including bottles), which were the most photo-resistant polymers studied. Based on the linear extrapolation of plastic mass loss, engineered polymer solutions (2.7 years) and the North Pacific Gyre (2.8 years) samples had the shortest lifetimes, followed by polypropylene (4.3 years), polyethylene (33 years), and standard polyethylene (49 years), used for crates, trays, bottles for milk and fruit juices, and caps for food packaging.¬Ý
“For the most photoreactive microplastics such as expanded polystyrene and polypropylene, sunlight may rapidly remove these polymers from ocean waters. Other, less photodegradable microplastics such as polyethylene, may take decades to centuries to degrade even if they remain at the sea surface,” said Shiye Zhao, Ph.D., senior author and a post-doc researcher working in the laboratory of , Ph.D., an assistant professor of biology/biogeochemistry at FAU’s Harbor Branch and . “In addition, as these plastics dissolve at sea, they release biologically active organic compounds, which are measured as total dissolved organic carbon, a major byproduct of sunlight-driven plastic photodegradation.”
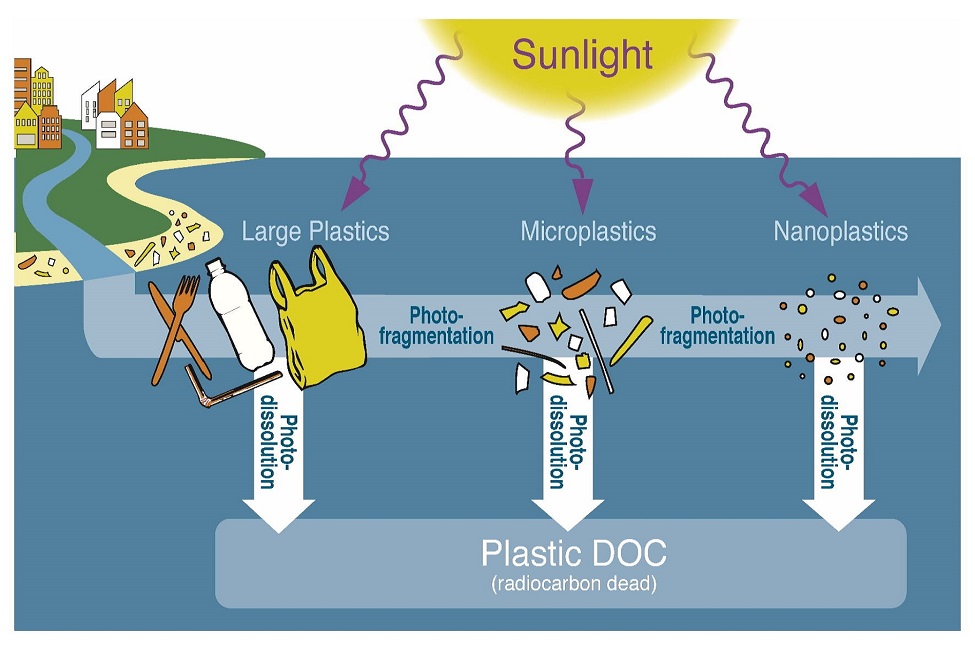
Zhao and collaborators also checked the biolability of plastic-derived dissolved organic carbon upon marine microbes. These dissolved organics seem to be broadly biodegradable and a drop in the ocean compared to natural biolabile marine dissolved organic carbon. However, some of these organics or their co-leachates may inhibit microbial activity. The dissolved organic carbon released as most plastics photodegraded was readily utilized by marine bacteria.
“The potential that plastics are releasing bio-inhibitory compounds during photodegradation in the ocean could impact microbial community productivity and structure, with unknown consequences for the biogeochemistry and ecology of the ocean,” said Zhao. “One of four polymers in our study had a negative effect on bacteria. More work is needed to determine whether the release of bioinhibitory compounds from photodegrading plastics is a common or rare phenomenon.”
Samples in the study included post-consumer microplastics from recycled plastics like a shampoo bottle and a disposable lunch box (polyethylene, polypropylene, and expanded polystyrene), as well as standard polyethylene, and plastic-fragments collected from the surface waters of the North Pacific Gyre. A total of 480 cleaned pieces of each polymer type were randomly selected, weighed and divided into two groups.
Co-authors of the study are Lixin Zhua, a Ph.D. student and lead author, East China Normal University; Thais B. Bittar, Ph.D.; and Aron Stubbins, Ph.D., both at Northeastern University; and Daoji Li, Ph.D., East China Normal University.
This work was supported by national key research and development program of China [2016YFC1402205], the United States National Science Foundation [1910621], The National Science Foundation of China [42676190, 41806137, 41676190), and a graduate fellowship from the Chinese Scholarship Council [201506140016].
-FAU-
Tags: harbor branch | technology | jupiter | science | faculty and staff | research | honors